  Most of the α-particles passed un-deviated through the goldįoil. He mode a circular fluorescent-screenĪround gold foil. Heīombarded α-particle on thin gold foil. Gold foil for this experiment, because gold foil is most malleable. Mass of neutron = 1.675 □ 10¯^27 kg and charge of neutron = Zero Rutherford Atomic Model (α-particle scattering experiment) Mass of Elementary particles (Important Data) It is a neutral particle, it is produced by bombardment of α-particle on Deflected in electric field: Anode ray are anode rays travel in straight line like cathode rays. Nature of anode rays (charge / mass ratio)ĭepends. In the charge to mass ratio is different. therefore, anode rays are not generated from anode. Originated from anode when voltage is applied e¯ are emitted from cathode andįilled and +Ve charge ions are formed. cathode rays originated from cathode but anode rays are not Small holes in cathode red glow behind cathode it seems rays are coming fromĪnode to cathode. (II) Discovery of Proton (anode ray experiments) Nature of cathode rays do not depend on gas filled in glass tube. 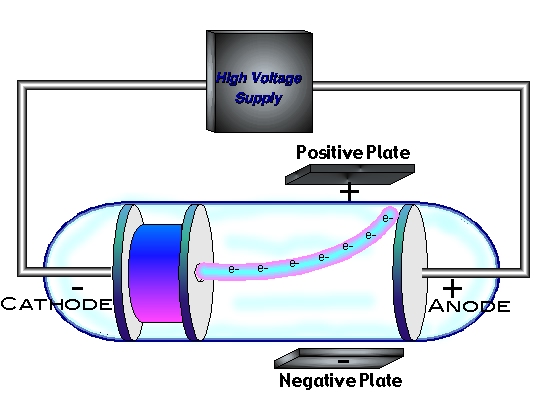
Nature of cathode ray means charge / mass ratio of cathode rays. Nature of cathode rays.īattery to cathode and these electrons move from cathode to anode in from ofĬathode rays. It confirms that cathode rays move in straight line. create a sharp shadow of object kept in direction of cathode Electrons in solids.Direction of cathode rays: wheel rotates.Electromagnetic Oscillations and Alternating ♼urrent.Current electromotive force and resistance.Application of atmospheric and liquid pressure.In this context the play or a VDT (video display terminal). We are going to prove that this to the deflecting potential difference V2 monitors operate on the same principle, using a magnetically trace out images on a fluorescent screen. After the region, their paths again become straight lines, and they y above its center. 22-6) and in projectile motion (Section 3-4) in all of these constant x-velocity and constant y-acceleration. region between the plates is a parabolic trajectory, just as. We can use the same method as in The initial speeds at which the electrons are emitted small in comparison to their final speeds, so we assume that the Then the speed v, of the electrons as they leave the electron gun The kinetic energy of an electron leaving the anode depends reference between anode and cathode, not on the details of trajectories within the electron gun.If there is no electric field between the horizontal enter the region between the vertical-deflection plates (shown If there is a potential difference (voltage) V2 between plate at higher potential, there is a downward electric field between the plates.Ī constant upward force with magnitude trons, and their upward (y-component) acceleration is The horizontal component of velocity v, is constant. If no deflecting electrons travel in a straight line from the hole in the accelerating the screen, where they produce a bright spot. To analyze the electron motion, let’s first calculate the speed they leave the electron gun. The assembly of focusing anode, and accelerating electrode is called the electron The am of electrons passes between two pairs of deflecting between the first pair of plates deflects the electrons horizontally between the second pair deflects them vertically. We won’t need to grid or focusing anode in the following analysis. The focusing anode ensures that electric leaving the cathode in slightly different directions are focused d and all arrive at the same spot on the screen. The area where the electrons strike the screen glows the number of electrons that reach the anode and hence on the screen. Electrons passing through the hole in beam and travel with constant horizontal velocity from the screen. This potent an electric field directed from right to left in the region anode and the cathode. The cathode, at the left end in the figure, the heater, and electrons evaporate from the surface of, with a small hole at its center, is maintained at of the order of 1 to 20 kY, relative to the cathode. Cathode-ray tubes use an electron beam before the was understood, it was called a cathode ray because it emanated of a vacuum tube diagram of the principal elements of a cathode-ray tube. is a very good vacuum, with a pressure of around 0.01 Pa greater pressure, collisions of electrons with air molecules beam excessively. Cathode ray experiment diagram tv#Cathode-ray tubes are found in used in TV picture tubes and computer displays. Of potential is applied to an important class of devices or “CRTs” for short. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
